| Mar 31, 2026 Elucidation of the Mechanisms Driving Pathogenicity of Synovial Macrophages |
Hiroki Mukoyama1,2, Yusuke Takeuchi1,2, Daiya Ohara1,3, Yoonha Lee1, Hitomi Watanabe1, Hiroki Kato4, Gen Kondoh1, Akio Morinobu2, Keiji Hirota1,4*
1) Laboratory of Integrative Biological Science, Institute for Life and Medical Sciences, Kyoto University, Kyoto, Japan.
2) Department of Rheumatology and Clinical Immunology, Graduate School of Medicine, Kyoto University, Kyoto, Japan
3) The Hakubi Center for Advanced Research, Kyoto University, Kyoto, Japan
4) Institute of Cardiovascular Immunology, Medical Faculty, University Hospital Bonn, University of Bonn, Bonn, Germany
Pathogenic GM-CSF drives functional diversification of inflammatory macrophages in autoimmune arthritis
Science Advances (2026) doi.org/10.1126/sciadv.aec0986
Abstract
Rheumatoid arthritis is an autoimmune disease affecting approximately 1% of the population, in which immune cells induce inflammation, leading to joint pain and swelling. Macrophages are a major source of inflammatory cytokines such as tumor necrosis factor (TNF) and play a central role in disease pathogenesis; in recent years, they have also been implicated in pain. Granulocyte–macrophage colony-stimulating factor (GM-CSF) has been shown in both mouse models and clinical trials to be deeply involved in the development of arthritis. However, the mechanisms by which macrophage-mediated inflammation and pain are regulated, as well as how GM-CSF acts on specific cell types to drive inflammation, have remained unclear. To address these questions, the research team investigated how GM-CSF affects synovial macrophages.
In this study, the researchers used SKG mice, a model of autoimmune arthritis that resembles human rheumatoid arthritis. First, they demonstrated that inflammatory monocytes are critical for arthritis development: CCR2-deficient SKG mice, in which inflammatory monocytes cannot migrate into the joints, exhibited significantly attenuated arthritis compared to wild-type SKG mice. Analysis of the development and differentiation of synovial macrophages revealed that circulating monocytes recruited from the bone marrow—rather than tissue-resident macrophages—serve as the primary precursors of pathogenic macrophages.
Next, mixed bone marrow chimeric mice were generated using CCR2-deficient SKG mice and GM-CSF receptor–deficient SKG mice. Blocking GM-CSF signaling in inflammatory monocytes and macrophages conferred resistance to arthritis, demonstrating that GM-CSF signaling is essential for the acquisition of pathogenic functions in these cells.
Furthermore, single-cell RNA sequencing of myeloid cells in inflamed synovial tissue was performed using mixed bone marrow chimeras of wild-type and GM-CSF receptor–deficient SKG mice. The analysis revealed that Arg1⁺ macrophages and EpCAM⁺ macrophages were absent among cells derived from GM-CSF receptor–deficient SKG mice. These macrophage subsets exhibited high expression of inflammatory cytokines, and notably, EpCAM⁺ macrophages specifically expressed CCL17, a molecule associated with pain. This study is the first to demonstrate that GM-CSF is indispensable for the differentiation of these functionally specialized, highly pathogenic macrophage populations.
These findings provide important insights into the mechanisms underlying arthritis pathogenesis and the link between inflammation and pain.
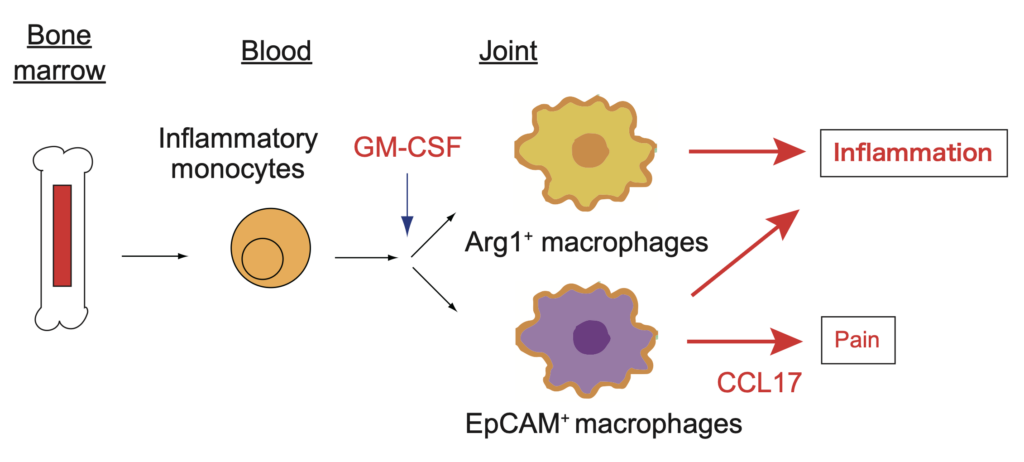
Figure) Inflammatory monocytes that migrate from the bone marrow into the bloodstream enter the joints, where they are exposed to GM-CSF and differentiate into Arg1⁺ and EpCAM⁺ macrophages, thereby contributing to the amplification of joint inflammation. In particular, EpCAM⁺ macrophages are also involved in pain through the production of CCL17.
